-
zopiclone impurities|SZEB
EP and BP contained zopiclone known impurities control A, B, C and 2-amino-5-chloropyridine, zopiclone raw materials in the destruction of the degradation of the situation, the light destructio...
See More
-
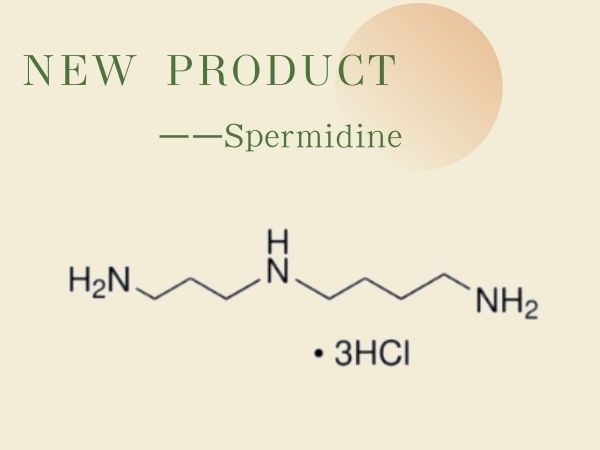
New Product--Spermidine | SZEB
Spermidine, also known as spermidine trihydrochloride, is a polyamine. Widely distributed in living organisms, it is biosynthesized from putrescine (butylenediamine) and adenosine methionine. SZE...
See More
-
Rhinitis Nemesis - Mometasone Nasal Spray 丨Mometasone impurity supply...
Mometasone furoate is a topical glucocorticoid that does not cause systemic effects at doses that exert local anti-inflammatory effects....
See More
-
Vancomycin, the last line of defense against infection...
Vancomycin is used primarily for infections of methicillin-resistant Staphylococcus aureus and other bacterial genera, and acts on bacteria by inhibiting bacterial cell wall synthesis, altering bact...
See More
-
Antibacterial and anti-inflammatory essential - Levofloxacin 丨SZEB...
Levofloxacin belongs to the quinolone class of antimicrobial drugs, and is commonly used for a variety of infections caused by sensitive bacteria in the respiratory system, urinary system, skin tissu...
See More
-
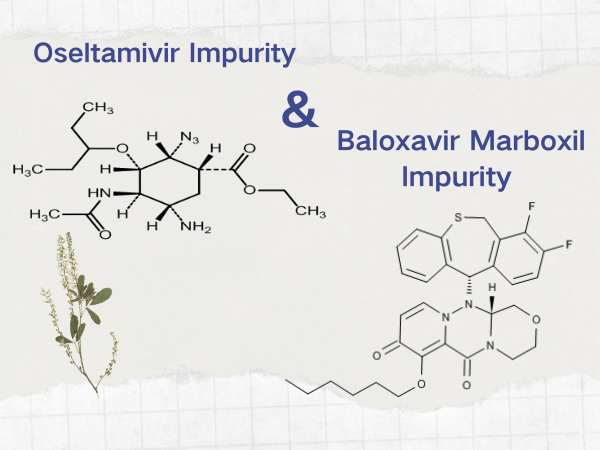
Marbofloxacavir and oseltamivir - anti-flu specialties 丨 SZEB...
It‘s high time for the flu again, and when it comes on strong.If you are not careful, you may have a sudden high fever, generalized weakness, cough and runny nose, limb aches and pains .........
See More
-
Paroxetine imprurites 丨SZEB
Paroxetine hydrochloride has antidepressant properties and is a selective 5-hydroxytryptamine reuptake inhibitor used in the treatment of neurological disorders such as depression, obsessive-compulsi...
See More
-
Orlistat Impurity 丨SZEB
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Orlistat related Impurity ,Discover...
See More
-
Isavuconazole Impurity | SZEB
Shenzhen Excellent Biomedical Technology Co., Ltd. (SZEB) is an innovative service enterprise focusing on the field of drug quality control, providing customers with impurity reference subst...
See More
-
Buy Nicardipine Impurity Online | SZEB
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Nicardipine related Impurity ,Disco...
See More
-
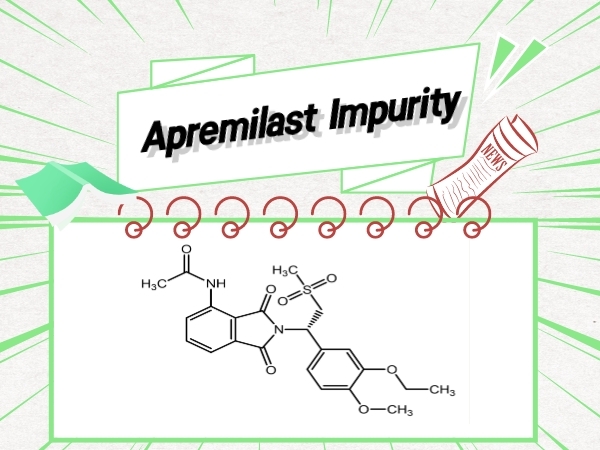
Psoriasis-Apremilast Impurity
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Apremilast related Impurity ,Discov...
See More
-
Upadacitinib - Drug Impurity | SZEB
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Upadacitinib related Impurity ,Disc...
See More
-
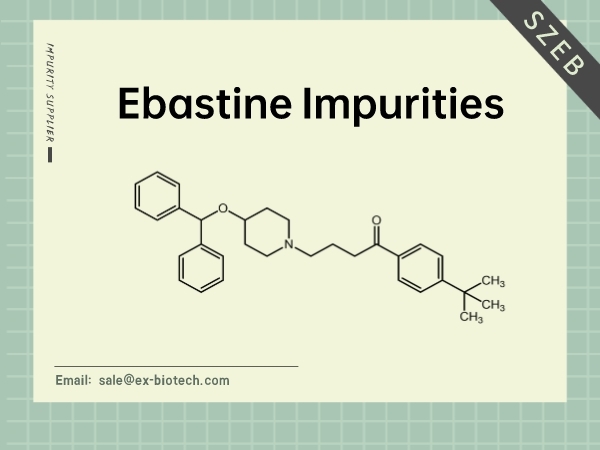
Anti-allergic Impurity-Ebastine
SZEB can provide the impurities of Ebastine -related impurities,Discover all our products through our official website (https://www.ex-biotech.com/), and get in touch if you have any questions ...
See More
-
Supply Pantoprazole Impurities | SZEB
SZEB can provide the impurities of Pantoprazole -related impurities,Discover all our products through our official website (https://www.ex-biotech.com/), and get in touch if you have any questi...
See More
-
Phlegm-Reducing Drug Impurities-Ambroxol
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Ambroxol related Impurity ,Discover...
See More
-
Sitagliptin - Pharmaceutical Impurities | SZEB...
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Sitagliptin related Impurity ,Disco...
See More
-
Diquafosol - Drug Impurity | SZEB
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Diquafosol related Impurity ,Discov...
See More
-
Immuno Suppressant Medication Impurity-Cyclosporin...
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Cyclosporin related Impurity ,Disco...
See More
-
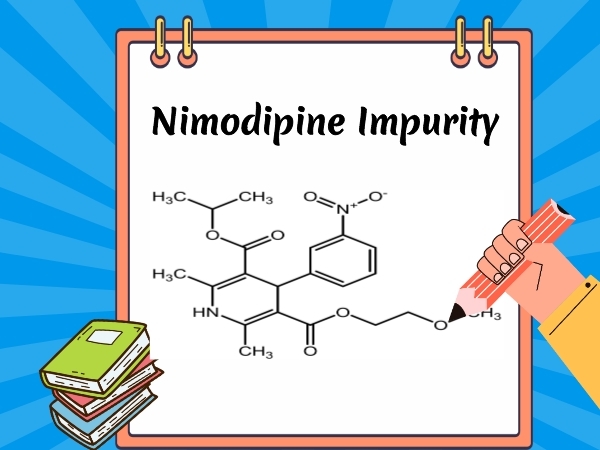
Nimodipine - Drug Impurity | SZEB
SZEB is a high-techbio-pharmaceutical company specializing in research,synthesis and supply of pharmaceutical reference standards independently..Supply of Nimodipine related Impurity ,Discov...
See More
-
Nebivolol-Drug Impurity-SZEB
We can provide various drug impurities,including starting materials, by-products, intermediates, transformation products, interaction products, degradation products, etc. Shenzhen Excellent Biotec...
See More



















.jpg) Wechat
Wechat